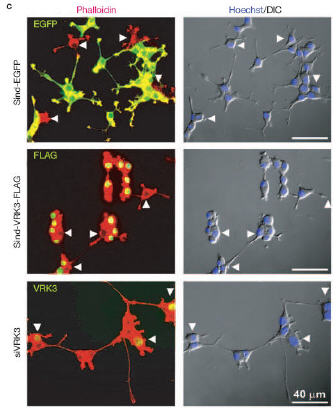
 | ||
[네이처지 일부]
Negative regulation of ERK activity by VRK3-mediated activation of VHR phosphatase
Tae-Hong Kang1 and Kyong-Tai Kim1,2
Extracellular signal regulated kinases (ERKs) represent a signalling hub in many hysiological responses and have pivotal functions in cell proliferation, differentiation, development and
death, as well as in synaptic plasticity1,2. Mitogen-activated protein kinase phosphatases [AU: Abbreviation must be spelled out in full at first use.
Please confirm that this is correct.](MKPs) selectively inactivate ERKs by dephosphorylating critical phosphothreonine and phosphotyrosine residues3,4. Transcriptional induction of MKP expression5,6 and posttranscriptional stabilization of MKP [AU: It is house style to italicize all genes, mRNA, siRNAs etc. OK?] mRNA7 are well-documented as negative-feedback mechanisms for ERK signalling. Vaccinia-related kinase 3 (VRK3) is a member of the novel VRK family8,9, but its function has not been defined.
Here, we show that VRK3 suppresses ERK activity through direct binding to one of the MKPs, vaccinia H1-related (VHR)10, which specifically dephosphorylates and inactivates ERK in the
nucleus11. Notably, VRK3 enhances the phosphatase activity of VHR by a mechanism independent of its kinase activity. VRK3 is therefore a member of a new class of hosphatase-activating kinases that regulate the activity of ERK. Our findings show that direct interaction of VHR with VRK3 posttranslationally regulates ERK signalling posttranslation [AU: OK? It is house style to avoid using the word ‘novel’]
뉴스타운
뉴스타운TV 구독 및 시청료 후원하기
뉴스타운TV

























![[특별대담] 제22대 국회의원 선거 자유민주당 인천 서구병 이진기 예비후보](/news/photo/202403/600022_545770_3853.jpg)






![[긴급시국분석] 북한의 내년 4월 총선 전후 기습남침과 좌파들의 내란 및 전민봉기 철저히 대비하라!](/news/thumbnail/202312/592978_536686_4219_v150.jpg)